- Home
- About Us
- Meet Norso

Career Opportunities
Join our team and collaborate with industry leaders to gain insights, experience and add value.
About Feature1
Who we are
Our expert team knows how to anticipate, collaborate, and innovate.
About feature2Get in Touch
Blackwater Business Park, Mallusk Way Newtownabbey, BT36 4AA
02896 005063 (UK)
01 531 4337 (ROI)
Mon-Fri, 9am until 5pmservice@norsomedical.co.uk
Contact feature
service@norsomedical.ie
We always reply within 24 hours
- Products
- Support
- News & Events
- Search
- Contact
- Home
- About Us
- Meet Norso

Career Opportunities
Join our team and collaborate with industry leaders to gain insights, experience and add value.
About Feature1
Who we are
Our expert team knows how to anticipate, collaborate, and innovate.
About feature2Get in Touch
Blackwater Business Park, Mallusk Way Newtownabbey, BT36 4AA
02896 005063 (UK)
01 531 4337 (ROI)
Mon-Fri, 9am until 5pmservice@norsomedical.co.uk
Contact feature
service@norsomedical.ie
We always reply within 24 hours
- Products
- Support
- News & Events
- Search
- Contact
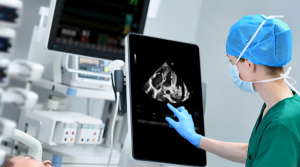
Norso Medical is committed to driving better patient outcomes by utilising cutting-edge technology. We specialise in the development of patient monitoring systems that provide clinicians with real-time data, allowing for informed decision-making that can ultimately improve patient care and outcomes.
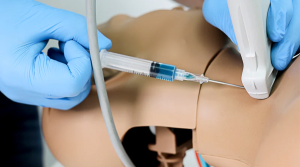
Norso Medical leverages simulation technology to deliver realistic, hands-on training to healthcare professionals. We partner with industry leaders like Monivent, Lifecast, and Gaumard to develop, to provide innovative simulation-based solutions that improve workforce training for hospitals, universities and emergency medical services.
Norso Medical provides hospital automation connectivity services, which involve the integration of various hospital systems and devices via a centralized network. This facilitates seamless communication and data exchange, ultimately leading to enhanced patient care, error reduction, improved efficiency, and optimized resource utilisation.
Improving Lives Together.
MINDRAY EPM10 | 12
Inspired by the needs of customers in the mid-acuity setting, the Mindray ePM patient monitor has been designed to meet the growing needs of caregivers, to enhance the efficiency and safety of the care they provide.
Mindray TEX20 Series
The Mindray TEX20 series ultrasound has an innovative new design that helps clinicians reimagine their clinical practice in demanding environments like critical care and emergency medicine.
Gaumard HAL® S5301
The S5301 sets the standard for the future of patient simulation, building upon the design philosophy established with the original HAL launched nearly two decades ago. Join us as we chart HAL’s course into the future.
Masimo UniView
The UniView system is an integrated display of real-time data and alarms that others its information from a number of Masimo and third-party devices.
Core Values: Driving Healthcare Excellence
United by a common set of shared values that guide our pursuit of healthcare excellence, innovation, and patient-centred care

Innovation
Innovation is at the core of our company ethos as a medical technology provider. We are committed to driving progress and creating new solutions that improve patient outcomes and enhance clinical practice.

Education
We prioritise education and training to empower healthcare professionals with the knowledge and skills needed to deliver optimal patient care and improve clinical outcome.

Sustainability
We recognise our responsibility to protect the environment and promote sustainability. We are committed to reducing our ecological impact and creating a healthier future for all.
Stay updated with our latest news, events and discover industry insights, product launches and recent white papers.
OUR PARTNERS
No matter how complex your business questions, we have the capabilities and experience to deliver the answers you need to move forward.